Rotarian and veterinarian Dr. Arnold L Goldman gave a presentation on the general subject of gene therapy for inherited disease, with the specific example of a muscle disorder found in male puppies and little boys called "X-Linked Myotubular Myopathy" or "XL-MTM". The presentation was based upon a more detailed presentation previously given by researcher Dr. David Mack of the University of Washington, Institute for Stem Cell & Regenerative Medicine, located in Seattle. Dr. Goldman had seen the presentation at the National Animal Interest Alliance Conference twice over the last few years and made it a point to meet the speaker.

The Joshua Frase Foundation helps to support research on XL-MTM, a disease that affects the X chromosome, the one chromosome that males possess just one copy of. Having no second X chromosome, males do not have an alternative set of potentially normal X chromosome genes to take over for those that might be defective on the first X chromosome. As females normally have two X chromosomes, and two copies of all X-linked genes, they can be carriers but are not affected by the disease.
In this case a gene that "codes" for an important muscle protein known as "myotubularin" is defective and little to no myotubularin is created. As a result, muscle cells, also known as fibers, in all the skeletal muscles of affected puppies and boys, remain in an undeveloped state, one which leads to extreme dysfunction.
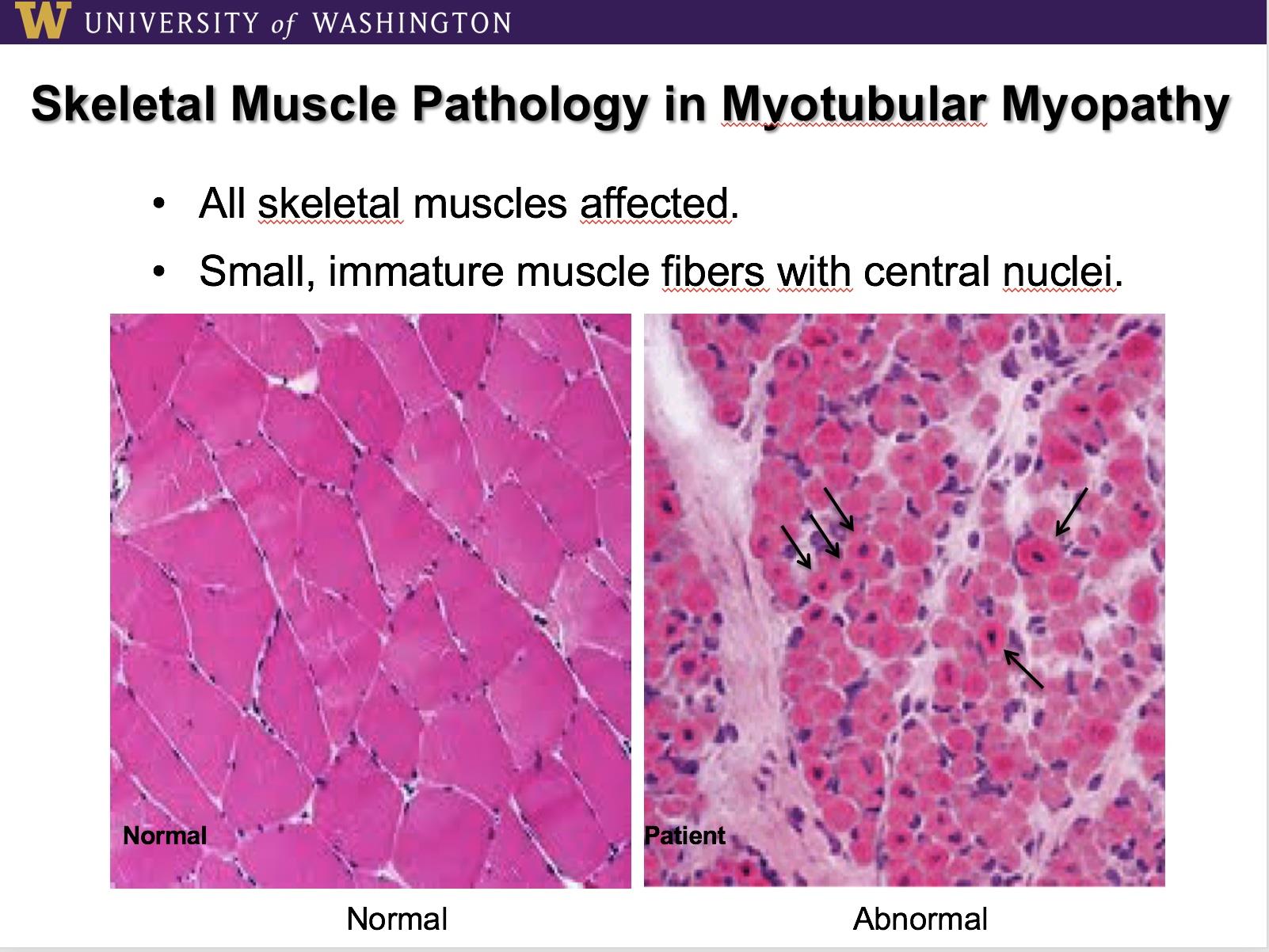
XL-MTM is a terrible disease with an eventually fatal outcome for 100% of affected patients. Worldwide the incidence is 1 in 50,000 live born, male infants, with boys born "floppy", with severe muscle weakness and a characteristic long narrow face. Greater than 50% will die before their 2nd birthday, with just a few surviving into their teens. Death is due to respiratory failure as the muscles of respiration are affected and eventually the compensatory mechanisms of the body are overwhelmed. In the case of the little boys, before they succumb, the care required of their parents and caregivers is extreme and most commonly includes 24-hour respiratory support with a ventilator, an intervention that is costly and labor intensive. That is in addition to, all the other needs of any growing child. The impact on families is devastating with a heavy burden of physical, emotional and financial consequences.
Alison Frase the heroic mother of one such boy, Joshua Frase, and also the wife of now-retired NFL player Paul Frase, would not be deterred in her efforts to help find a way to save her son Joshua, who was affected by the disease. With encouragement from Joshua's doctors, she scoured the nation, and the world, for similar disease victims among other species, so that they might be studied in the hunt for a treatment for her son.

Mrs. Frase learned from a San Diego veterinary pathologist about a litter of puppies that had died shortly after being born, and with similar signs. The mother dog was owned by a Saskatchewan farm family, and Alison flew to Saskatoon that very night, arriving unannounced at the farm house doorstep after midnight. After explaining who she was, and that her son might have the same inherited illness as the farmer's dog, the farmer gave her the dog, named Nibs, to bring to the scientists to study. Alison returned to North Carolina with Nibs and soon put Nibs into the University of Washington researcher's hands.

The dog lived with the researcher's family in Seattle and not long after arriving, Nibs gave birth to another litter of 12 puppies. This litter included 1 affected male and 5-affected female carriers, and thus the study colony, and the critical research, was begun. As the research progressed, healthy puppies were placed with the human patient's families, so that they could have a personal connection to the research.
Barely 1 year later, one of the many collaborators in this research, Dr. Alan Beggs of Harvard University, published the results of a study identifying the gene "MTM1" as the gene that was defective in Nibs, her puppies and the affected little boys, including young Joshua Frase.
The next step was to pursue a treatment. The strategy was to develop a method to insert a normal copy of the MTM1 gene into abnormal muscle cells, causing the protein myotubularin to be produced, and then hope muscle structure and function would return to normal. To do that, a benign virus that normally targets muscle was used to deliver a normal MTM1 gene into cells that lack one. The process first depends on selecting a virus with a natural affinity for muscle, like the kind that causes muscle aches, so the functional gene "cargo" is delivered where it needs to go, and can be absorbed by the cell's own mechanisms into its chromosomes.
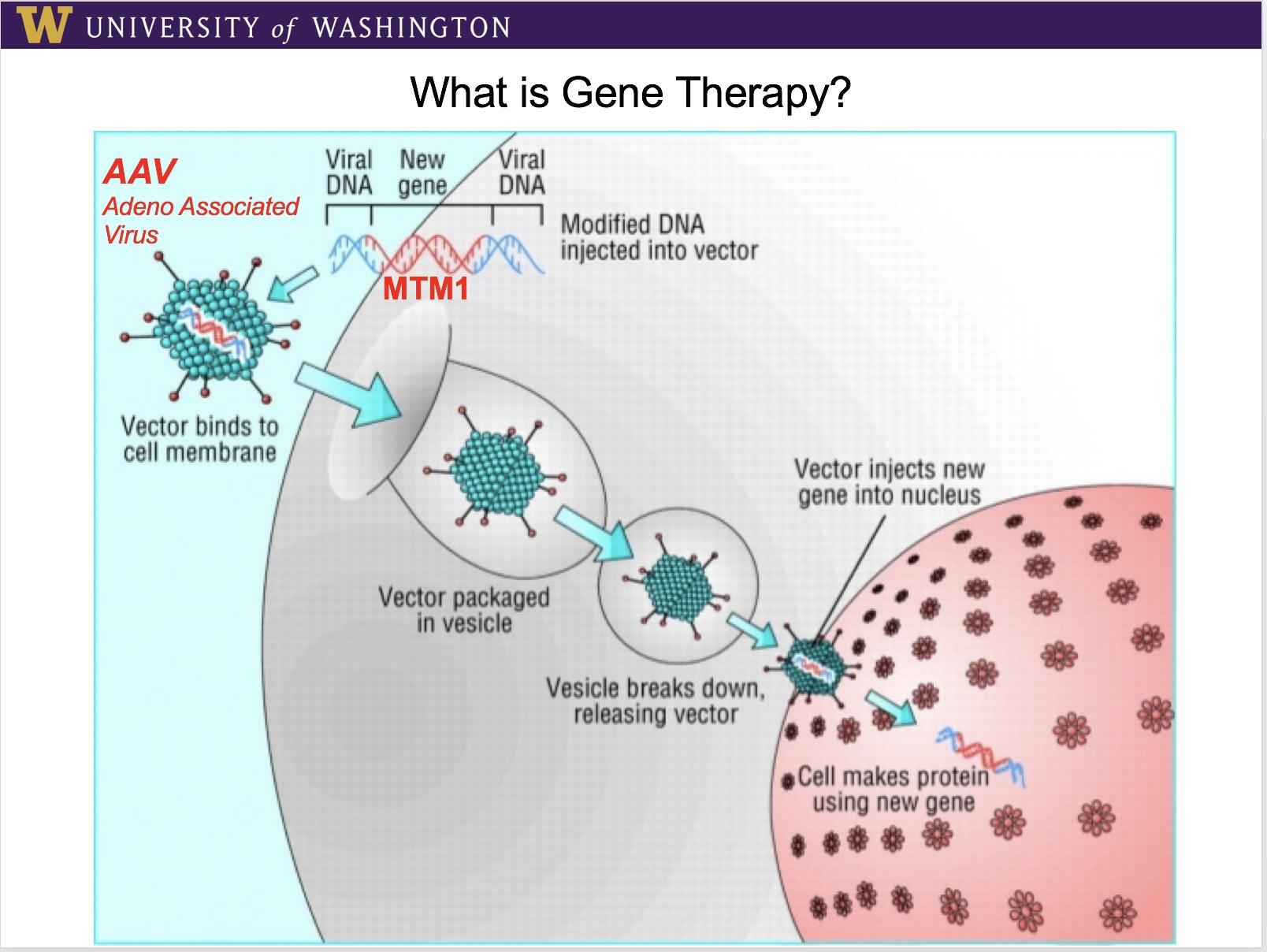
This method of viral gene delivery was first tried in mice engineered to have the defective MTM1 gene, and then in the next phase, in naturally affected dogs. The results in the dogs were more successful than could possibly have been imagined, with prolonged survival, restored muscle strength, reversed structural abnormalities, and no adverse immune system reactions to the virus or gene, and all that after a single treatment! What's more, 5 years on, the first two treated dogs remain healthy.
After publishing the successful outcomes in the treated dogs, the scientists approached the FDA for permission to treat the little boys, and soon received it. At a minimum it would be transformative if this therapy could restore even some respiratory function to these boys.
Dr. Goldman showed Rotarians video of young, affected dogs both before and after receiving this gene therapy. While for privacy and legal reasons he was unable to show us similar video of untreated & treated boys, he informed the audience that at least three boys have since been treate. He reported that all three are vastly improved and for the first time, functioning normally. Many other such boys are now in line for similar treatment in the near future.

Some important questions remain, questions that gene therapy scientists have yet to answer. Will one dose last for the lifetime of a patient?; Will the virus persist in muscle & cause trouble later?; In infants, does therapy weaken as the baby grows to an adult?; If one dose doesn’t suffice, can the exact same virus/gene combination be administered again without adverse consequences?; Will there then be immune reactions to the virus, the gene or both?; Will the virus persist & evolve?; Will that be harmful?; Clearly, Dr. Goldman said, there is so much more work remaining to be done.

He continued, saying "often in veterinary practice, I'm asked what I think about the use of animals in biomedical research, for example beagle dogs, and whether its unethical to use them." "I tell those who've asked, that research using animals benefits both people & animals and is essential to solve many health problems that animals and humans share. These so-called, animal “models” of human disease are essential for progress to be made, as there is no inanimate substitute for this kind of research. Computer simulations and tissue culture have their uses, but cannot model a living organism in all its complexity. Further, so called “rare” diseases often have common characteristics, sometimes allowing similar approaches to study & treatment. Finally, precisely because this research saves human lives, not doing it is what would be unethical."
Dr. Goldman's final point was that it had cost 18 million dollars a month for 5 years, to get to this point with
XL-MTM. There are dozens of other diseases suitable for this form of treatment that are being studied with lives at stake. Please support and promote biomedical research, and also the Joshua Frase Foundation.